
Medical communicators new to writing clinical study reports may find the process overwhelming. The report is more detailed and structured than many other regulatory documents, and new report writers don’t necessarily know what regulators or stakeholders expect.
What's the best approach to writing a clinical study report (CSR)? Medical writers typically develop these documents in collaboration with a team, following a general structure that includes a synopsis, an introduction, processes, outcomes, discussion, and conclusions.
This blog covers the basics for medical communicators tasked with preparing clinical study reports, whether as part of regulatory submissions, investigator-initiated studies, or publication support. By the end of this article, you’ll understand what a clinical study report is, when it's needed, the essential components of the report, and the team members involved, giving you a roadmap to approach clinical study report writing with confidence.
October 6, 2025 at 4:00 AM




































 One of the most useful acronyms in a medical communicator’s toolkit is
One of the most useful acronyms in a medical communicator’s toolkit is 
 Artificial intelligence (AI) is transforming how medical research is conducted and how clinical trials are designed, run, and analyzed. From accelerating data analysis to improving participant recruitment, AI is helping researchers and clinicians bring treatments to patients faster and more effectively.
Artificial intelligence (AI) is transforming how medical research is conducted and how clinical trials are designed, run, and analyzed. From accelerating data analysis to improving participant recruitment, AI is helping researchers and clinicians bring treatments to patients faster and more effectively.


 If you are just getting started in medical writing and don’t have a portfolio, taking that step can boost your credibility and help you advance your career. Yet, the idea of building a medical writing portfolio can be overwhelming.
If you are just getting started in medical writing and don’t have a portfolio, taking that step can boost your credibility and help you advance your career. Yet, the idea of building a medical writing portfolio can be overwhelming.
 Apprenticeships are an age-old institution used to guide people in their careers and help them develop the skills they need to succeed. AMWA has an ongoing interest in exploring the value of apprenticeships and similar training programs, including hosting the
Apprenticeships are an age-old institution used to guide people in their careers and help them develop the skills they need to succeed. AMWA has an ongoing interest in exploring the value of apprenticeships and similar training programs, including hosting the 
 As a regulatory writer, you must stay updated on new policies and evolving best practices for highly technical documents. Continuing education is important for both maintaining and advancing your
As a regulatory writer, you must stay updated on new policies and evolving best practices for highly technical documents. Continuing education is important for both maintaining and advancing your 
 As generative artificial intelligence (AI) tools—like ChatGPT and Gemini—evolve, freelance medical writers are striving to balance new capabilities with professional responsibilities. Whether you're cautiously exploring or fully embracing generative AI tools, knowing how to use them ethically and effectively is critical. With some key insights, AI can become a powerful ally to increase efficiency and build capacity in your freelance medical writing business.
As generative artificial intelligence (AI) tools—like ChatGPT and Gemini—evolve, freelance medical writers are striving to balance new capabilities with professional responsibilities. Whether you're cautiously exploring or fully embracing generative AI tools, knowing how to use them ethically and effectively is critical. With some key insights, AI can become a powerful ally to increase efficiency and build capacity in your freelance medical writing business.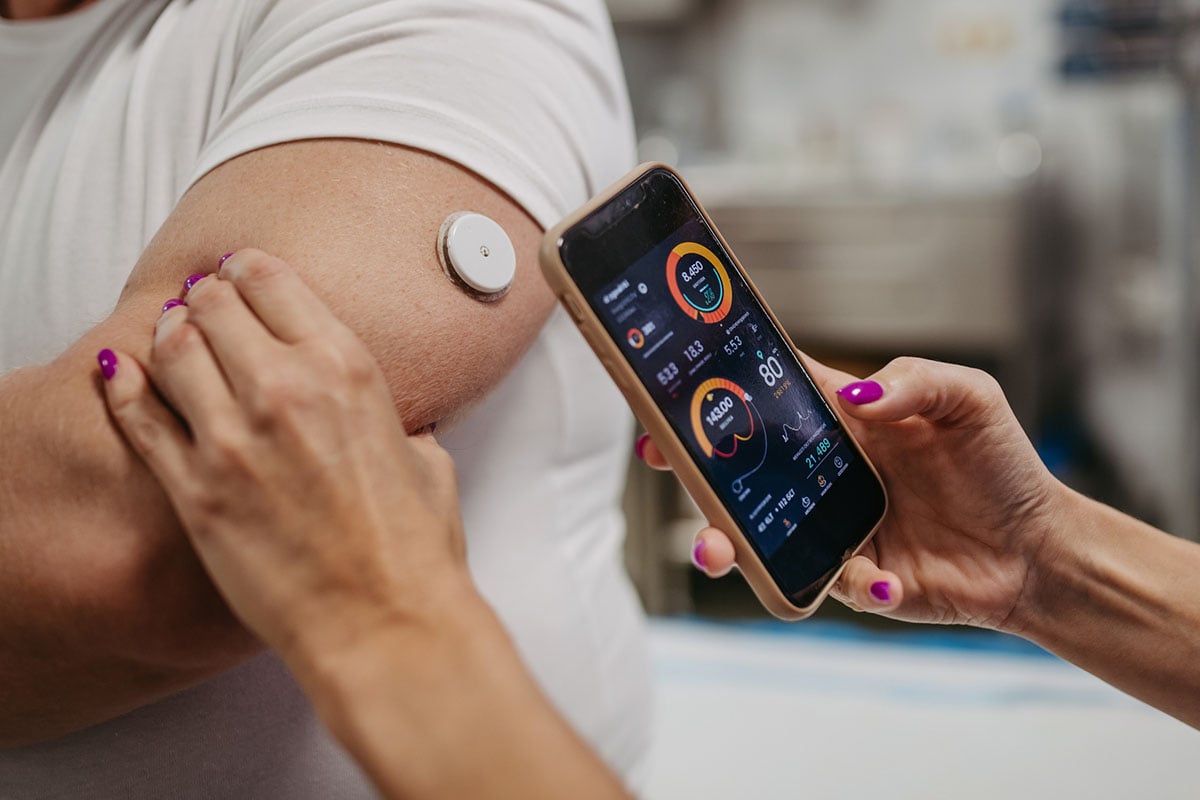
 Medical communicators who work in the pharmaceutical space will recognize some features of medical device writing.
Medical communicators who work in the pharmaceutical space will recognize some features of medical device writing.
 Inequities related to income, race, education, language, and age can affect access to quality health care, health and wellness outcomes, and health literacy. Medical communicators play a crucial role communicating complex medical and scientific data and creating accessible health care content for various audiences.
Inequities related to income, race, education, language, and age can affect access to quality health care, health and wellness outcomes, and health literacy. Medical communicators play a crucial role communicating complex medical and scientific data and creating accessible health care content for various audiences.
 Note: The tips here are meant to provide an overview of best practices regarding business setup for freelance medical communicators. For advice specific to your situation, seek counsel from authorized financial experts.
Note: The tips here are meant to provide an overview of best practices regarding business setup for freelance medical communicators. For advice specific to your situation, seek counsel from authorized financial experts.
 As artificial intelligence (AI) transforms the digital landscape, medical writers and health communication professionals must adapt their content strategies to stay relevant in search engine optimization (SEO). AI-driven search algorithms are reshaping the way health care content is ranked and discovered online. In this evolving environment, content must be clear and concise while balancing the need for accuracy in health communication.
As artificial intelligence (AI) transforms the digital landscape, medical writers and health communication professionals must adapt their content strategies to stay relevant in search engine optimization (SEO). AI-driven search algorithms are reshaping the way health care content is ranked and discovered online. In this evolving environment, content must be clear and concise while balancing the need for accuracy in health communication.
 Regulatory writing
Regulatory writing
 Millions of people log in to one or more social media sites every day, and patients and families are increasingly seeking health information online. This merging of digital media and health care realms makes it imperative that health care organizations, clinicians, researchers, and medical communicators engage with audiences on social media channels.
Millions of people log in to one or more social media sites every day, and patients and families are increasingly seeking health information online. This merging of digital media and health care realms makes it imperative that health care organizations, clinicians, researchers, and medical communicators engage with audiences on social media channels. 
 Medical writers and editors are often the core contributors on writing projects, and they need a solid understanding of grammar, punctuation, usage, and style.
Medical writers and editors are often the core contributors on writing projects, and they need a solid understanding of grammar, punctuation, usage, and style.
 Regulatory medical writers are key personnel in delivering quality regulatory documents that advance science and medicine, keeping patients safe and informed about the benefits and risks of pharmaceutical products.
Regulatory medical writers are key personnel in delivering quality regulatory documents that advance science and medicine, keeping patients safe and informed about the benefits and risks of pharmaceutical products. 
 In recent years, the medical writing landscape has evolved to include a crucial new voice: the patient. As patient-centered care and shared decision-making gain momentum, patients are increasingly contributing to medical literature, advocacy materials, and even peer-reviewed research.
In recent years, the medical writing landscape has evolved to include a crucial new voice: the patient. As patient-centered care and shared decision-making gain momentum, patients are increasingly contributing to medical literature, advocacy materials, and even peer-reviewed research.
 Every medical communicator finds their own path to success.
Every medical communicator finds their own path to success.
 Can medical communicators share an article from a medical journal with colleagues? What about reusing a graph from a published paper? What is fair use, and how do you know the difference between noncommercial and commercial use for licensing?
Can medical communicators share an article from a medical journal with colleagues? What about reusing a graph from a published paper? What is fair use, and how do you know the difference between noncommercial and commercial use for licensing?
 Medical communicators and publications share a serious responsibility for advancing scientific knowledge and following ethical practices and guidelines.
Medical communicators and publications share a serious responsibility for advancing scientific knowledge and following ethical practices and guidelines. 
 Artificial intelligence (AI) and the subcategory machine learning (ML) are evolving quickly, and regulators around the world are working to respond to new opportunities while mitigating potential risks involving the technology.
Artificial intelligence (AI) and the subcategory machine learning (ML) are evolving quickly, and regulators around the world are working to respond to new opportunities while mitigating potential risks involving the technology. 
 With so much digital content vying for our attention, medical communicators need to harness the power of visual communication. Otherwise, high-quality health care messaging could end up buried in a sea of social media reels and misleading posts.
With so much digital content vying for our attention, medical communicators need to harness the power of visual communication. Otherwise, high-quality health care messaging could end up buried in a sea of social media reels and misleading posts. 
 Are you looking for a career pivot from the world of research to medical writing?
Are you looking for a career pivot from the world of research to medical writing?


 To succeed as a medical communicator, writers and editors must master essential technical skills such as grammar, medical terminology, and statistics. However,
To succeed as a medical communicator, writers and editors must master essential technical skills such as grammar, medical terminology, and statistics. However, 
 Health is much more than numbers, medications, or a clinical diagnosis. A person’s health is affected by their environment and their lived experiences.
Health is much more than numbers, medications, or a clinical diagnosis. A person’s health is affected by their environment and their lived experiences. 




 Health care or medical public relations (PR) is a growing niche for medical communicators that involves translating and sharing complex medical information with a general audience of patients and consumers.
Health care or medical public relations (PR) is a growing niche for medical communicators that involves translating and sharing complex medical information with a general audience of patients and consumers. 
 Clinical consensus statements and clinical practice guidelines are among the documents used to ensure that patients are receiving the best care possible and that clinical decisions reflect the opinions of the experts who are invested in excellent clinical outcomes.
Clinical consensus statements and clinical practice guidelines are among the documents used to ensure that patients are receiving the best care possible and that clinical decisions reflect the opinions of the experts who are invested in excellent clinical outcomes.




 Every document improves with editing, and medical editors are key to the process of creating consistent, readable, and error-free manuscripts.
Every document improves with editing, and medical editors are key to the process of creating consistent, readable, and error-free manuscripts.






 What’s your style? Are you an AMA person? An APA aficionado? Does your employer use a house style guide?
What’s your style? Are you an AMA person? An APA aficionado? Does your employer use a house style guide? 
 This blog is based, in part, on an AMWA webinar presented by Monica Nicosia, PhD, CMPP.
This blog is based, in part, on an AMWA webinar presented by Monica Nicosia, PhD, CMPP.
 We do not live in a world where everyone — regardless of race, gender, gender identity, disability status, or income level — has the same access to high-quality health care, adequate housing, or nutrition. As a result, health outcomes are vastly unequal, causing marginalized populations to experience lower life expectancies and higher levels of preventable diseases.
We do not live in a world where everyone — regardless of race, gender, gender identity, disability status, or income level — has the same access to high-quality health care, adequate housing, or nutrition. As a result, health outcomes are vastly unequal, causing marginalized populations to experience lower life expectancies and higher levels of preventable diseases.
 So much of our lives is connected online these days, and freelance medical communicators are using tools that allow collaboration with colleagues around the world.
So much of our lives is connected online these days, and freelance medical communicators are using tools that allow collaboration with colleagues around the world. 
 We live in an increasingly globalized world, and the field of medical communication is no exception. In fact, most medical writers and editors are creating materials for a global scientific audience.
We live in an increasingly globalized world, and the field of medical communication is no exception. In fact, most medical writers and editors are creating materials for a global scientific audience.


 Are you a regulatory medical writer who is concerned about proving your mettle?
Are you a regulatory medical writer who is concerned about proving your mettle?
 Scientists are having a rough go of it right now. They are being harassed at record levels, especially for sharing public health information related to the COVID-19 pandemic.
Scientists are having a rough go of it right now. They are being harassed at record levels, especially for sharing public health information related to the COVID-19 pandemic. Are you prepared for the next generation of science communication?
Are you prepared for the next generation of science communication?


 What role do medical communicators play in the world of health care reimbursement?
What role do medical communicators play in the world of health care reimbursement? 
 As health care costs continue to rise, medical communicators can play a role in health care policy and practice by participating in Health Economics and Outcomes Research (HEOR)—a growing field with opportunities for medical communicators.
As health care costs continue to rise, medical communicators can play a role in health care policy and practice by participating in Health Economics and Outcomes Research (HEOR)—a growing field with opportunities for medical communicators. 


 Randomized controlled trials (RCTs) are the foundation for advancing clinical practice. However, not all RCTs are created equal. To communicate how an RCT was designed and what it found, authors must report clear and accurate information about its methodology and results. Unfortunately, many reviews have shown that authors often don’t provide complete and transparent information about the clinical trial, which can lead to misinterpretation of the validity and reliability of the results.
Randomized controlled trials (RCTs) are the foundation for advancing clinical practice. However, not all RCTs are created equal. To communicate how an RCT was designed and what it found, authors must report clear and accurate information about its methodology and results. Unfortunately, many reviews have shown that authors often don’t provide complete and transparent information about the clinical trial, which can lead to misinterpretation of the validity and reliability of the results. 


 If patients and consumers don’t understand patient education materials — or don't know what action to take after reading or viewing them — those materials aren’t doing their job.
If patients and consumers don’t understand patient education materials — or don't know what action to take after reading or viewing them — those materials aren’t doing their job.
 Storytelling is the basis for human connection, with oral and written traditions stretching back for millennia.
Storytelling is the basis for human connection, with oral and written traditions stretching back for millennia. 
 By Stephen N. Palmer, PhD, ELS, Texas Heart Institute
By Stephen N. Palmer, PhD, ELS, Texas Heart Institute


 By Laura J. Ninger, ELS, Ninger Medical Communications, LLC
By Laura J. Ninger, ELS, Ninger Medical Communications, LLC




 By Charlotte Crowder, MPH, ELS, Freelance Medical Writer and Editor
By Charlotte Crowder, MPH, ELS, Freelance Medical Writer and Editor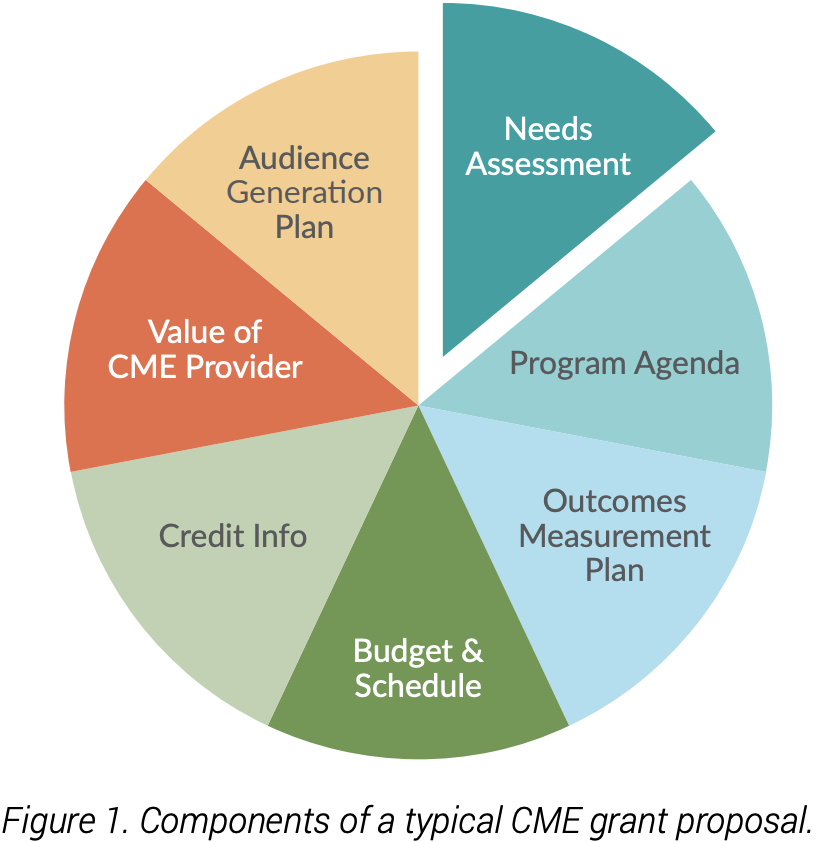

 Are you taking time for self-care?
Are you taking time for self-care?
 Copyright law is complicated, and we’re not legal experts.
Copyright law is complicated, and we’re not legal experts.


 Those in the medical writing and editing field know that job responsibilities vary widely.
Those in the medical writing and editing field know that job responsibilities vary widely. 
 Ethical behavior is important in any profession.
Ethical behavior is important in any profession. 
 Checklists are popular these days. And for good reason.
Checklists are popular these days. And for good reason. 
 The American Medical Writers Association (AMWA) has selected Leana Wen, MD, MSc, FAAEM, an emergency physician, public health professor at George Washington University, and nonresident senior fellow at the Brookings Institution, as the 2022 Walter C. Alvarez Award Recipient. This award recognizes excellence in communicating health care developments and concepts to the public and is named after Walter C. Alvarez, MD, a pioneer in the field of medical communication.
The American Medical Writers Association (AMWA) has selected Leana Wen, MD, MSc, FAAEM, an emergency physician, public health professor at George Washington University, and nonresident senior fellow at the Brookings Institution, as the 2022 Walter C. Alvarez Award Recipient. This award recognizes excellence in communicating health care developments and concepts to the public and is named after Walter C. Alvarez, MD, a pioneer in the field of medical communication.
 The American Medical Writers Association (AMWA) has selected Peter J. Hotez, MD, PhD, an internationally recognized physician-scientist, global health advocate, accomplished author, and 2022 Nobel Peace Prize nominee, as the 2022 John P. McGovern Award Recipient. This award recognizes preeminent contributions to the various modes of medical communication and is named in honor of renowned allergist and philanthropist John P. McGovern, MD.
The American Medical Writers Association (AMWA) has selected Peter J. Hotez, MD, PhD, an internationally recognized physician-scientist, global health advocate, accomplished author, and 2022 Nobel Peace Prize nominee, as the 2022 John P. McGovern Award Recipient. This award recognizes preeminent contributions to the various modes of medical communication and is named in honor of renowned allergist and philanthropist John P. McGovern, MD.
 Have you taken a stretch break in the last 15 minutes? Do it! Lift your arms up above your head and tilt back slightly to counteract that forward curve. Yes, we’re talking to you.
Have you taken a stretch break in the last 15 minutes? Do it! Lift your arms up above your head and tilt back slightly to counteract that forward curve. Yes, we’re talking to you.
 Your time as a medical writer is worth money.
Your time as a medical writer is worth money. 
 This article is based on content presented by
This article is based on content presented by 
 Have you been asked to sign a confidentiality agreement as part of a medical writing or editing
Have you been asked to sign a confidentiality agreement as part of a medical writing or editing 
 This blog is based on content presented by Kathi Whitman, MA, Senior Medical Writer/Project Manager, Intermountain Healthcare, at the AMWA Medical Writing & Communication Conference and published in AMWA Journal v 31 n 4.
This blog is based on content presented by Kathi Whitman, MA, Senior Medical Writer/Project Manager, Intermountain Healthcare, at the AMWA Medical Writing & Communication Conference and published in AMWA Journal v 31 n 4. 
 People take different paths in their journeys to become medical writers. Some are scientists and clinicians who come to the field with medical or scientific backgrounds. Others are professional writers who want to leverage their background in communications to write about science and medicine.
People take different paths in their journeys to become medical writers. Some are scientists and clinicians who come to the field with medical or scientific backgrounds. Others are professional writers who want to leverage their background in communications to write about science and medicine. 


 The hunt for freelance medical writers and editors begins when you have a need for a communication professional who can help you complete or improve your writing project. Sometimes you have an urgent need, and you don’t know where to turn.
The hunt for freelance medical writers and editors begins when you have a need for a communication professional who can help you complete or improve your writing project. Sometimes you have an urgent need, and you don’t know where to turn.


 How do medical communicators distill the results of complex studies into just a few hundred words?
How do medical communicators distill the results of complex studies into just a few hundred words? 
 Imagine a toolbox without a hammer.
Imagine a toolbox without a hammer. 
 An essential skill for medical communicators involved in creating materials for continuing medical education (CME) activities is learning to identify a practice gap by conducting a gap analysis.
An essential skill for medical communicators involved in creating materials for continuing medical education (CME) activities is learning to identify a practice gap by conducting a gap analysis.
 How are you with numbers?
How are you with numbers?
 This blog is based on content presented by
This blog is based on content presented by 


 Freelance success is possible. As today’s work landscape rapidly changes, more talented professionals are leaving traditional employment settings, or are interested in supplementing their income, and are finding value in the flexibility and economic opportunities presented by freelance work.
Freelance success is possible. As today’s work landscape rapidly changes, more talented professionals are leaving traditional employment settings, or are interested in supplementing their income, and are finding value in the flexibility and economic opportunities presented by freelance work. 
 One niche for medical writers is grant writing. Whether they are writing grant proposals to fund continuing medical education (CME) programs or to support biomedical research, grant writers are tasked with convincing funders that the project is needed now, and that their team is the best one to undertake the project on time and within budget.
One niche for medical writers is grant writing. Whether they are writing grant proposals to fund continuing medical education (CME) programs or to support biomedical research, grant writers are tasked with convincing funders that the project is needed now, and that their team is the best one to undertake the project on time and within budget. 
 This blog is based on an article written by
This blog is based on an article written by 
 When people are looking to establish or further a career as a medical writer, they often have questions about the differences between earning a certificate in medical writing and certification, which means earning the Medical Writer Certified (MWC®) credential.
When people are looking to establish or further a career as a medical writer, they often have questions about the differences between earning a certificate in medical writing and certification, which means earning the Medical Writer Certified (MWC®) credential.
 As a medical writer, chances are high that you’ve crafted a document or two without your byline attached. After all, much of the content medical writers create doesn’t require authorship. If you’re interested in
As a medical writer, chances are high that you’ve crafted a document or two without your byline attached. After all, much of the content medical writers create doesn’t require authorship. If you’re interested in 
 This article is based on content presented by
This article is based on content presented by 


 If you’re a
If you’re a 


 If you’re a
If you’re a 
 As a freelance medical writer, you must master a variety of business skills in addition to writing high‑quality documents. You’ll need to
As a freelance medical writer, you must master a variety of business skills in addition to writing high‑quality documents. You’ll need to 
 In Entrepreneur’s May 2020 article “
In Entrepreneur’s May 2020 article “
 Whether you're hoping to expand your portfolio to include medical journal writing or are already incorporating journal articles into your
Whether you're hoping to expand your portfolio to include medical journal writing or are already incorporating journal articles into your 
 If there’s one key to success as a freelance medical writer, it’s this: never stop marketing.
If there’s one key to success as a freelance medical writer, it’s this: never stop marketing. 
 The economic fallout from the COVID‑19 pandemic has affected thousands of career paths, including many in the medical communication field. Whether you’re seeking new remote work opportunities or hoping to expand your skill set to further
The economic fallout from the COVID‑19 pandemic has affected thousands of career paths, including many in the medical communication field. Whether you’re seeking new remote work opportunities or hoping to expand your skill set to further 
 The medical communication field is expanding at a record pace. At the same time, remote work is becoming mainstream. By demand, many experienced
The medical communication field is expanding at a record pace. At the same time, remote work is becoming mainstream. By demand, many experienced 
 If you’re a new medical writer, you may wonder which skills are essential for success. Whether you choose to write regulatory documents, promotional content, patient education materials, or journal articles, you’ll need a strong writing foundation. Part of that foundation includes learning to understand, write, speak—in general, to communicate!—using the jargon of the field: medical terminology.
If you’re a new medical writer, you may wonder which skills are essential for success. Whether you choose to write regulatory documents, promotional content, patient education materials, or journal articles, you’ll need a strong writing foundation. Part of that foundation includes learning to understand, write, speak—in general, to communicate!—using the jargon of the field: medical terminology. 




 This article by Jodi Braunton was originally published in the
This article by Jodi Braunton was originally published in the 
 This article, written by
This article, written by 
 Amid ongoing global health issues and a shifting economy, many experienced
Amid ongoing global health issues and a shifting economy, many experienced 
 This article is based on content presented by
This article is based on content presented by 


 This article is based on content presented by
This article is based on content presented by 
 Note: The tips here are meant to provide an overview of best practices regarding accounting needs for freelancers. For advice specific to your situation, seek counsel from authorized financial experts.
Note: The tips here are meant to provide an overview of best practices regarding accounting needs for freelancers. For advice specific to your situation, seek counsel from authorized financial experts.


 Many medical writers are tasked with more than crafting accurate documents. You might also need to manage projects and teams.
Many medical writers are tasked with more than crafting accurate documents. You might also need to manage projects and teams. 


 To work in health communication, you need both detailed knowledge about health care and marketing and the ability to manage a variety of editorial and writing tasks.
To work in health communication, you need both detailed knowledge about health care and marketing and the ability to manage a variety of editorial and writing tasks.
 For freelance medical writers and editors, social media can help build authority, engage with colleagues, connect with potential clients, and grow their freelance businesses. But posting and sharing inconsistently, or using the wrong platforms, can waste time and energy. These social media marketing tips can help.
For freelance medical writers and editors, social media can help build authority, engage with colleagues, connect with potential clients, and grow their freelance businesses. But posting and sharing inconsistently, or using the wrong platforms, can waste time and energy. These social media marketing tips can help.


 For some medical writers, a next step in their careers is a move to management. While this can create opportunities for advancement and greater recognition, it can be challenging—many medical writers are seasoned communicators, but they may not yet have the skills required to manage others.
For some medical writers, a next step in their careers is a move to management. While this can create opportunities for advancement and greater recognition, it can be challenging—many medical writers are seasoned communicators, but they may not yet have the skills required to manage others.
 This article is based on content written by Kelly Schrank, MA, ELS, and presented by John West Hadley at the AMWA Medical Writing & Communication Conference. It was originally published in the AMWA Journal.
This article is based on content written by Kelly Schrank, MA, ELS, and presented by John West Hadley at the AMWA Medical Writing & Communication Conference. It was originally published in the AMWA Journal.
 All of us have suffered the consequences of expensive, unasked questions in both our professional lives and our personal lives. As medical communicators, we must ask good questions to elicit information, but many of us lack adequate training in this skill. Add to that the natural reticence of some
All of us have suffered the consequences of expensive, unasked questions in both our professional lives and our personal lives. As medical communicators, we must ask good questions to elicit information, but many of us lack adequate training in this skill. Add to that the natural reticence of some 
 This article is based on content presented by
This article is based on content presented by 
 This article is based on content presented by
This article is based on content presented by 
 As medical writers, the heart of our work is to convey information to a specific audience. Writing for patients, we are trusted with interpreting highly technical information to educate, protect, enable, and empower our readers. The result of poor understanding can be directly harmful to health. However, successfully informing patients leads to better health outcomes, including more preventative care and
As medical writers, the heart of our work is to convey information to a specific audience. Writing for patients, we are trusted with interpreting highly technical information to educate, protect, enable, and empower our readers. The result of poor understanding can be directly harmful to health. However, successfully informing patients leads to better health outcomes, including more preventative care and 
 This article is based on content presented by
This article is based on content presented by 
 This article is based on content presented by
This article is based on content presented by 
 What is a servant leader? Servant leaders know their employees as whole people and strive to be a positive force in their lives. They build a first-hand understanding of the workplace. They create teams, address disparity, and create a vision for the growth of individuals and business. While most leaders are considered bosses or supervisors, all of us can serve as leaders to our colleagues and to peers as well. To start the path to leadership, it’s important to understand the qualities needed to act as a servant leader.
What is a servant leader? Servant leaders know their employees as whole people and strive to be a positive force in their lives. They build a first-hand understanding of the workplace. They create teams, address disparity, and create a vision for the growth of individuals and business. While most leaders are considered bosses or supervisors, all of us can serve as leaders to our colleagues and to peers as well. To start the path to leadership, it’s important to understand the qualities needed to act as a servant leader.
 I don’t make my living as a writer; let’s get that out of the way right now. However, I recruit writers and managers/directors of groups of medical writers, and I’ve been a mentor to many people. I’ve seen people succeed and fail, overcome obstacles and get back up again. A mentor or two (or more) can catapult an average career into a stellar career if you align yourself as early as possible with people who will be in your corner as you navigate your future.
I don’t make my living as a writer; let’s get that out of the way right now. However, I recruit writers and managers/directors of groups of medical writers, and I’ve been a mentor to many people. I’ve seen people succeed and fail, overcome obstacles and get back up again. A mentor or two (or more) can catapult an average career into a stellar career if you align yourself as early as possible with people who will be in your corner as you navigate your future.
 This article was written by
This article was written by
 It's fun to look back over the years and think about fashion trends that probably never should have seen the light of day. I’m thinking mullets, shoulder pads, and why not throw in bell bottoms while I’m at it.
It's fun to look back over the years and think about fashion trends that probably never should have seen the light of day. I’m thinking mullets, shoulder pads, and why not throw in bell bottoms while I’m at it.
 Recruiters can make all the difference when advancing your medical writing career. Experienced recruiters can help you identify a role that increases your earning potential, improve your work/life balance, and move you up to a senior-level position.
Recruiters can make all the difference when advancing your medical writing career. Experienced recruiters can help you identify a role that increases your earning potential, improve your work/life balance, and move you up to a senior-level position.
 Writing and editing go hand-in-hand to create a masterpiece. A well-written article is like a fine piece of jewelry. Think of the content as the design of the jewelry and the editing as the polish. Just as a ring without polish looks like a piece of lackluster metal with a potentially beautiful gem, a poorly edited article loses its charm even if the content is strong.
Writing and editing go hand-in-hand to create a masterpiece. A well-written article is like a fine piece of jewelry. Think of the content as the design of the jewelry and the editing as the polish. Just as a ring without polish looks like a piece of lackluster metal with a potentially beautiful gem, a poorly edited article loses its charm even if the content is strong.

 In medical communication, as in other fields, it’s increasingly common for team members to be scattered across the country or around the globe. It’s important for managers to have the skills needed to make their remote teams successful—no matter where employees are located. Managers of remote medical communication teams face challenges including
In medical communication, as in other fields, it’s increasingly common for team members to be scattered across the country or around the globe. It’s important for managers to have the skills needed to make their remote teams successful—no matter where employees are located. Managers of remote medical communication teams face challenges including
 A medical writing career requires being a lifelong learner and networker.
A medical writing career requires being a lifelong learner and networker.


 Did you know that today we receive 5 times as much information as we did in 1986? This amount is the equivalent of
Did you know that today we receive 5 times as much information as we did in 1986? This amount is the equivalent of